Neurodevelopmental disorders
Modelling pre-natal and early-life risk factors: (MIA) and cannabis
Neurodevelopmental and psychiatric disorders such as schizophrenia and autism spectrum disorder (ASD) are thought to arise due to exposure to a combination of pre- and postnatal risk factors. Epidemiological evidence has consistently identified in utero exposure to maternal immune activation (MIA) as a risk factor for these neurodevelopmental disorders. The gestational timing of MIA-exposure may influence the type and severity of neurodevelopmental disruptions in offspring since neurodevelopmental processes and maternal cytokine responsiveness vary across gestation.

To address these questions, we examine the impact of early or late prenatal maternal immune activation (MIA) on brain development throughout the lifespan using mice as a model system. We examine the effects of MIA on embryo and neonatal brain development using high resolution ex vivo structural MRI; we also examine the effects of early or late MIA on developmental trajectories from childhood to adulthood in mice using longitudinal whole-brain magnetic resonance imaging (MRI), multi-behavioural characterization, multivariate methods and transcriptional profiling. We found that early exposure to MIA led to the greatest brain-behaviour alterations, particularly in the adolescent period, but that these differences normalize in adulthood.
Building on this work, we have expanded the investigation of early-life risk factors into new neuroimaging modalities incorporating not only structural MRI and behaviors, but also magnetic resonance spectroscopy, to non-invasively assess chemical composition of brain tissue and functional MRI to query connectivity. Using multivariate techniques, we strive to understand not only how risk factors impact these modalities individually, but how the findings relate to form a deep characterization of the developing brain in health and adversity.
Figure : Mouse embryo at gestational day 18. T2-weighted, Gadolinium enhanced ex vivo scan (40um3 resolution; acquired at the Mouse Imaging Center, Toronto)
To better understand the vulnerability observed in the adolescent period due to MIA, we examine whether exposure to a second (postnatal) risk factor during the adolescent period leads to long-lasting changes, namely, adolescent cannabis exposure. Prospective, longitudinal, epidemiological studies consistently report an association between pre-psychosis cannabis use and schizophrenia after controlling for other drugs. In future work, we will use multimodal assessments to explore the effects of prenatal and adolescent THC exposure on brain and behavior over the course of early life.

Neuroanatomy in individuals at high risk for and suffering from a first episode of psychosis
The goal of this study is to investigate risk factors and psychobiological markers for psychosis in different groups at varying levels of risk for developing psychosis. We are recruiting individuals who have suffered a first episode of psychosis (FEP), individuals who are at high risk (HR) for developing psychosis because they have started to experience some symptoms (clinical high risk or CHR), individuals who are at risk because they have a sibling who has suffered an episode of psychosis (familial high risk or FHR), and healthy individuals (without family history). Using a longitudinal study design, these subjects are assessed at four time points over 1.5 years. We evaluate neuroanatomy using high resolution MRI and sophisticated techniques to detect subtle microstructural alterations in both grey and white matter, as well as cognitive and symptom profiles in order to characterise a variety of biomarkers that could aid in the early identification and diagnosis of psychotic disorders. We are now also measuring the impact of physical distancing restrictions imposed during the COVID-19 pandemic on stress, cognitive functions, and the brain using a self-report questionnaire, along with the brain measures. Our primary objective is to determine whether abnormalities in these psychobiological markers in HR and FEP subjects increase along with risk state (FHR < CHR < FEP), are specific to either HR or FEP, or are shared across familial and nonfamilial groups (regardless of HR or FEP status). Ultimately, this will improve our ability to identify risk markers and offer appropriate, targeted interventions.
Treatment resistant vs non-treatment resistant schizophrenia

Figure : Z-scored measures of Fractional Anisotropy (FA), Magnetic Resonance Spectroscopy (MRS) and Cortical Thickness (CT) for Traitment Resistant (TRS) and Traitment Non-Resistant (TnRS) schizophrenia patients, and Healthy Controls (HC).
Approximately 20 to 40% of patients with schizophrenia experience a limited response to first-line (i.e. non-clozapine) antipsychotics and are thus deemed to have treatment-resistant schizophrenia (TRS). Given the lack of effective available therapeutic options for patients with TRS, an improved understanding of its etiopathogenesis is of critical importance. For this, a multimodal neuroimaging examination of patients with TRS alongside patients who respond to first-line antipsychotics [i.e. treatment non-resistant schizophrenia (TnRS)] and healthy controls (HCs) is warranted given that, to the best of our knowledge, no previous study has concomitantly examined multiple imaging modalities in patients with TRS. In a participant sample composed of patients with TRS, patients with TnRS, and HCs, data was collected from multiple neuroimaging modalities, including structural MRI, diffusion tensor imaging, and proton magnetic resonance spectroscopy. Using multivariate approaches such as non-negative matrix factorization and partial least squares analysis, we are concomitantly investigating these multiple neuroimaging assays and assessing their association with demographics, clinical symptoms, and functional and cognitive impairment in this patient population.
Neuroanatomy of
autism spectrum disorders
To date, there is a lack of robust neurobiological markers that can guide diagnostic efforts and personalized treatment approaches in autism spectrum disorder (ASD). Progress has been impeded by inconsistencies in the neuroimaging literature, due in part to modest sample sizes, variability in subject selection, methodological differences, and the limitations of common statistical approaches. This study therefore aims to apply novel computational models that combine multiple morphometric data types in a large sample to improve our understanding of the neuroanatomy of ASD and its relation to clinical variability.
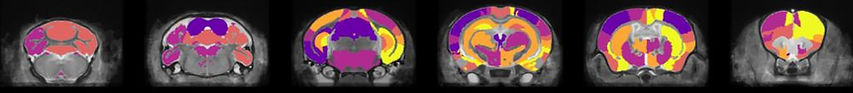
The impact of litter variability in longitudinal studies
.jpg)
Mouse models are commonly used in neuroscientific research studying both normative and disordered brain development. However, our current understanding of within- and between-litter variability in brain anatomy and behavioral measures may limit the translation of findings. Improved understanding of this variability can be used to carefully plan experiments and account for the trade-off between sample size and effect size. This is particularly important as a litter-effect could easily subsume a biological effect given the greater reported variance of measures across than within litters. These observations may be attributable to developmental factors such as shared maternal care, intrauterine and postnatal environment. Further, there is no clear understanding of the impact of litter variability in brain anatomy or behavioural measures. To date, there is no consensus in the analysis approaches used to account for the litter-specific variance. Improper accounting for this litter-effect may lead to increases in the reporting of true or false positives, further limiting translational and reproducibility of findings in neurodevelopmental research. We aim to better understand this litter variability through the observation of normal behavior and brain anatomy development in mouse models. We hope that a better understanding of this variability will lead to an improved understanding of experimental mouse models and consequently to the potential for improved translation in humans.
.jpg)
